Electrochemical Decarboxylative Hydroxylation for the Sustainable Synthesis of Bempedoic Acid
Abstract
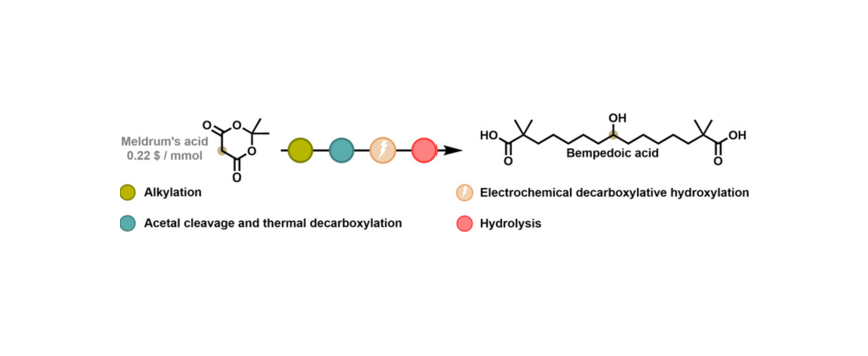
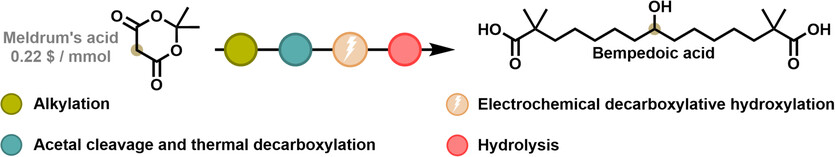
Bempedoic acid is a clinically approved lipid-lowering drug that inhibits adenosine triphosphate-citrate lyase, offering an alternative to statins for patients with statin intolerance or inadequate low-density lipoprotein cholesterol reduction. However, existing synthetic routes rely on hazardous reagents and complex multistep procedures. Here, we report a sustainable and efficient synthesis of bempedoic acid based on electrochemical hydroxylative decarboxylation as the key transformation. Starting from Meldrum's acid, an optimized sequence of alkylation, hydrolysis, and decarboxylation affords the carboxylic acid precursor under mild conditions and in the absence of metal-containing reagents. Subsequent electrochemical oxidation in dimethylformamide/hexafluoroisopropanol, using graphite and stainless-steel electrodes, enables direct conversion of the acid to the corresponding alcohol intermediate in up to 60% yield, followed by base-promoted hydrolysis to yield bempedoic acid. Compared with previously reported TosMIC- and malonate-based routes, this approach eliminates the use of toxic and pyrophoric reagents such as NaH and Pd/C, while improving atom economy and scalability. The method described herewith represents a robust and practical synthetic strategy for the synthesis of bempedoic acid and highlights the potential of electrochemical methodologies for sustainable pharmaceutical manufacturing.Pietro Ronco,Antonia Simi,Enrico Lunghi,Federico Della Negra,Beatrice Trucchi,Massimo Verzini,Giuseppe ZanoniPietro Ronco,Antonia Simi,Enrico Lunghi,Federico Della Negra,Beatrice Trucchi,Massimo Verzini,Giuseppe Zanoni