The Laurencia Stereochemical Paradox: Chemical Shift Litmus Test, Asymmetric Total Synthesis, and Structural Reassignment of (+)-Itomanallene B
Ervis Saraci, Federico Barbieri, Federica Plavi, Alessio Porta, Emanuele Casali and Giuseppe Zanoni
Abstract
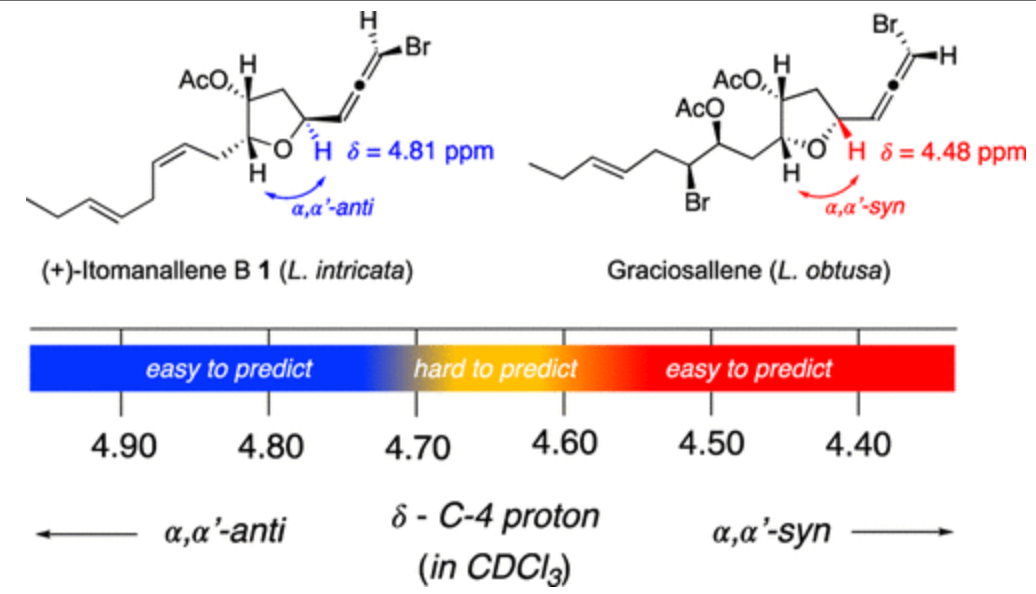
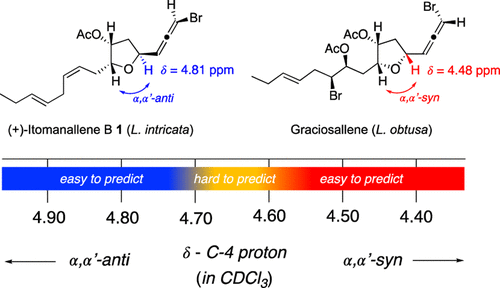
Red algae of the genus Laurencia are prolific sources of halogenated C15-acetogenins, many of which contain a tetrahydrofuran (THF) ring and a bromoallene terminus. Among them, Itomanallene B-type metabolites have presented a stereochemical paradox, as Laurencia intricata and Laurencia nangii yield epimeric structures despite sharing the same planar connectivity. In this study, we demonstrate that the chemical shift of the diagnostic C-4 proton provides a rapid and reliable indicator of the relative orientation (α,α′-syn vs α,α′-anti) of the THF side chains. Comparative analysis of reported NMR data reveals that anti configurations exhibit systematically more deshielded C-4 proton resonances than their syn counterparts. Applying this “chemical-shift litmus test” shows that Itomanallene B 1 from L. intricata possesses an α,α′-anti arrangement, whereas the metabolite previously reported as Itomanallene B 3 from L. nangii is instead the α,α′-syn epimer, 4-epi-Itomanallene B. This reassignment was unambiguously confirmed through the total asymmetric synthesis of the (aS,4S,6R,7R) stereoisomer of (+)-Itomanallene B, whose NMR data matched those of the natural product. The simplicity and diagnostic power of the C-4 proton chemical-shift approach provide an efficient tool for the stereochemical assignment of THF-containing C15-acetogenins, addressing a recurrent source of structural misinterpretation within the Laurencia genus.Ervis Saraci, Federico Barbieri, Federica Plavi, Alessio Porta, Emanuele Casali and Giuseppe Zanoni